Absolute entropy of ideal gas4/10/2024 
Processes for which S < 0 are not possible. 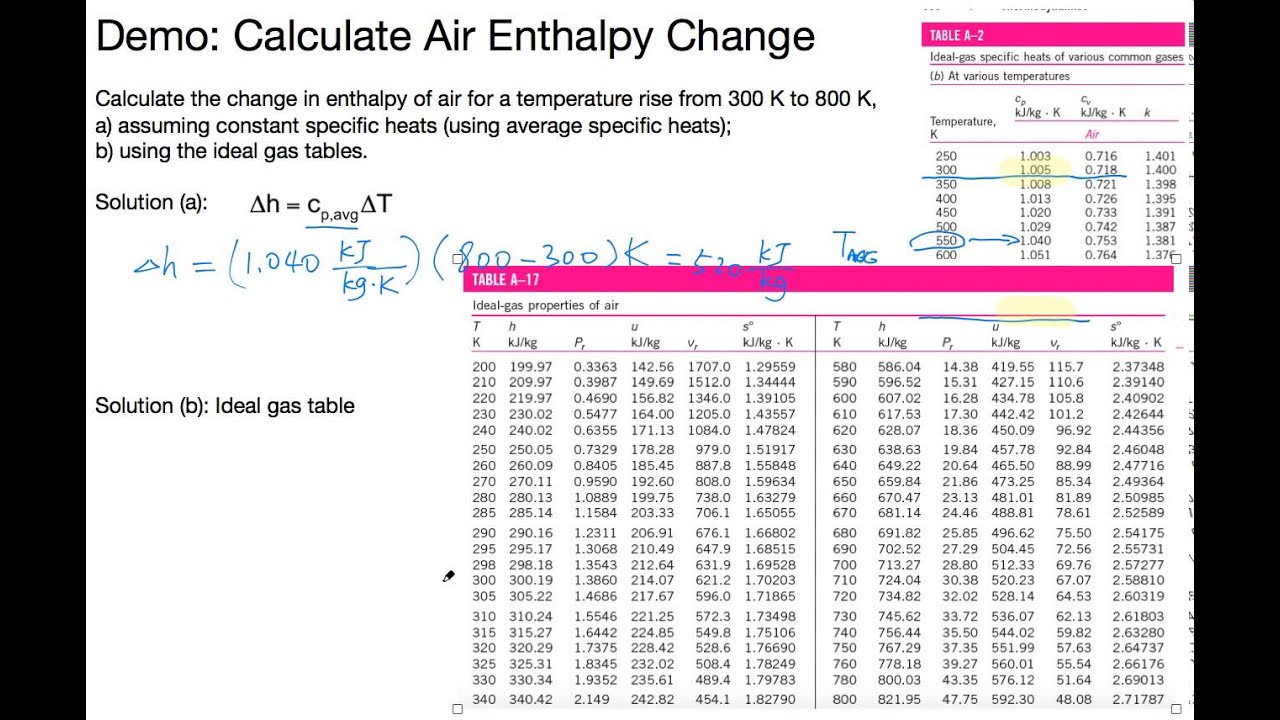
The second law states that the only direction for spontaneous changes in the entropy is an increase. It is a statement, not about the system alone, but about the full thermodynamic universe. Since the ideal gas law neglects both molecular size and intermolecular attractions, it is most accurate for monatomic gases at high temperatures and low pressures.\hspace-.6pt\raise1. Equation 12.65 is known as the second law of thermodynamics. There are in fact many different forms of the equation of state. The equation of state given here ( PV = nRT) applies only to an ideal gas, or as an approximation to a real gas that behaves sufficiently like an ideal gas. Third law: The entropy of a perfect crystal is zero when the temperature of the crystal is equal to absolute zero (0 K). Deviations from ideal behavior of real gases For reference, the Joule–Thomson coefficient μ JT for air at room temperature and sea level is 0.22 ☌/ bar. This is known as the Joule–Thomson effect. 
For real gasses, the molecules do interact via attraction or repulsion depending on temperature and pressure, and heating or cooling does occur. In the case of free expansion for an ideal gas, there are no molecular interactions, and the temperature remains constant. In an isenthalpic process, system enthalpy ( H) is constant. In internal combustion engines γ varies between 1.35 and 1.15, depending on constitution gases and temperature. Also γ is typically 1.6 for mono atomic gases like the noble gases helium (He), and argon (Ar). The value used for γ is typically 1.4 for diatomic gases like nitrogen (N 2) and oxygen (O 2), (and air, which is 99% diatomic). Under these conditions, p 1 V 1 γ = p 2 V 2 γ, where γ is defined as the heat capacity ratio, which is constant for a calorifically perfect gas. In an isentropic process, system entropy ( S) is constant. diverges at the ideal gas (or ideal solid). In the final three columns, the properties ( p, V, or T) at state 2 can be calculated from the properties at state 1 using the equations listed. A proper reference system and efficient switching path are necessary for accurate computatIOn of absolute entropy. Also, the property for which the ratio is known must be distinct from the property held constant in the previous column (otherwise the ratio would be unity, and not enough information would be available to simplify the gas law equation). The fundamental particles of nature have minimum vibrational motion, retaining only. Absolute zero is the lowest limit of the thermodynamic temperature scale a state at which the enthalpy and entropy of a cooled ideal gas reach their minimum value, taken as zero kelvin. We start with the entropy S(T,V)S(T0,V0) 3nR 2 log T T0 +nRlog V V0 3nR 2 log U U0 +nRlog V V0 of the ideal gas, as derived in Sect. Zero kelvin (273.15 ☌) is defined as absolute zero. We now use these relations to derive an expression for U in terms of its natural variables. 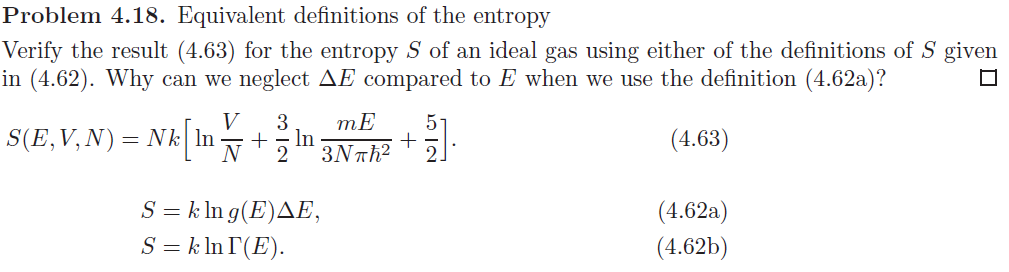
As shown in the first column of the table, basic thermodynamic processes are defined such that one of the gas properties ( P, V, T, S, or H) is constant throughout the process.įor a given thermodynamics process, in order to specify the extent of a particular process, one of the properties ratios (which are listed under the column labeled "known ratio") must be specified (either directly or indirectly). 5.1.1 Monoatomic ideal gas For the monoatomic ideal we did nd hitherto PV nRT, U 3 2 nRT, CV 3 2 nR. This function handles the retrieval of a chemicals absolute entropy at a reference temperature of 298.15 K and pressure of 1 bar, in the ideal gas state. The table below essentially simplifies the ideal gas equation for a particular processes, thus making this equation easier to solve using numerical methods.Ī thermodynamic process is defined as a system that moves from state 1 to state 2, where the state number is denoted by subscript. Here we will introduce a simplified method for calculating the change of. Where p Īpplications to thermodynamic processes The SackurTetrode equation is an expression for the entropy of a monatomic classical ideal gas, which incorporates quantum considerations that give a more. The specific entropy of an ideal gas is a function of both temperature and pressure.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
