|
While Mendeleev's periodic table and the modern periodic table have many similarities, there are also notable differences between the two. Lastly, both tables have contributed significantly to the advancement of chemistry and our understanding of the natural world. Thirdly, both tables serve as a foundation for further scientific research and experimentation, guiding the discovery of new elements and the exploration of their properties. Secondly, they both provide a systematic way to understand the behavior of elements and predict their chemical reactions. Firstly, both tables organize elements into periods and groups, highlighting the recurring patterns in their properties. Similaritiesĭespite their differences, Mendeleev's periodic table and the modern periodic table share several fundamental attributes. Lastly, the modern periodic table incorporates the concept of electron configuration, which helps explain the periodicity of elements and their chemical behavior. Thirdly, it includes the lanthanides and actinides as separate rows at the bottom, ensuring a more comprehensive representation of all known elements. Secondly, the modern periodic table has a more precise organization of elements into periods and groups, allowing for a better understanding of trends and relationships. Firstly, it is based on atomic number rather than atomic mass, providing a more accurate representation of an element's properties. The modern periodic table has several attributes that distinguish it from Mendeleev's table. The modern periodic table arranges elements in order of increasing atomic number, with elements having similar properties placed in the same vertical columns, known as groups or families. It was developed over time by various scientists, most notably Henry Moseley, who established the concept of atomic number in 1913. The modern periodic table, also known as the long form periodic table, is an updated version that incorporates advancements in our understanding of atomic structure and properties. Lastly, his periodic table provided a foundation for further research and experimentation, leading to the discovery of new elements and the refinement of the periodic table itself. Thirdly, Mendeleev's table left gaps for undiscovered elements, demonstrating his foresight and ability to predict their properties.  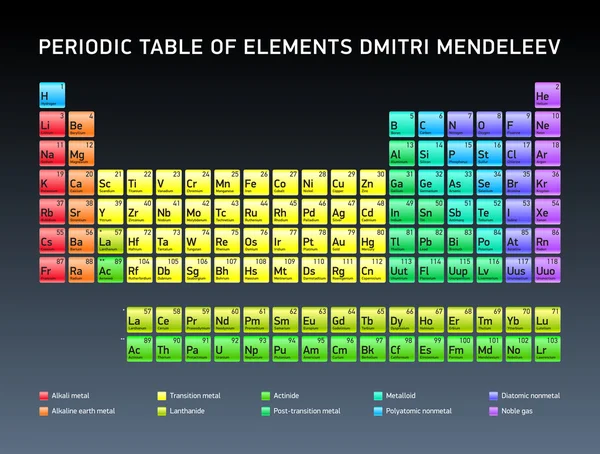
Secondly, it grouped elements with similar properties together, allowing for the identification of trends and patterns. Firstly, it organized elements based on their atomic mass, which was the best available information at the time. Mendeleev's periodic table had several key attributes. 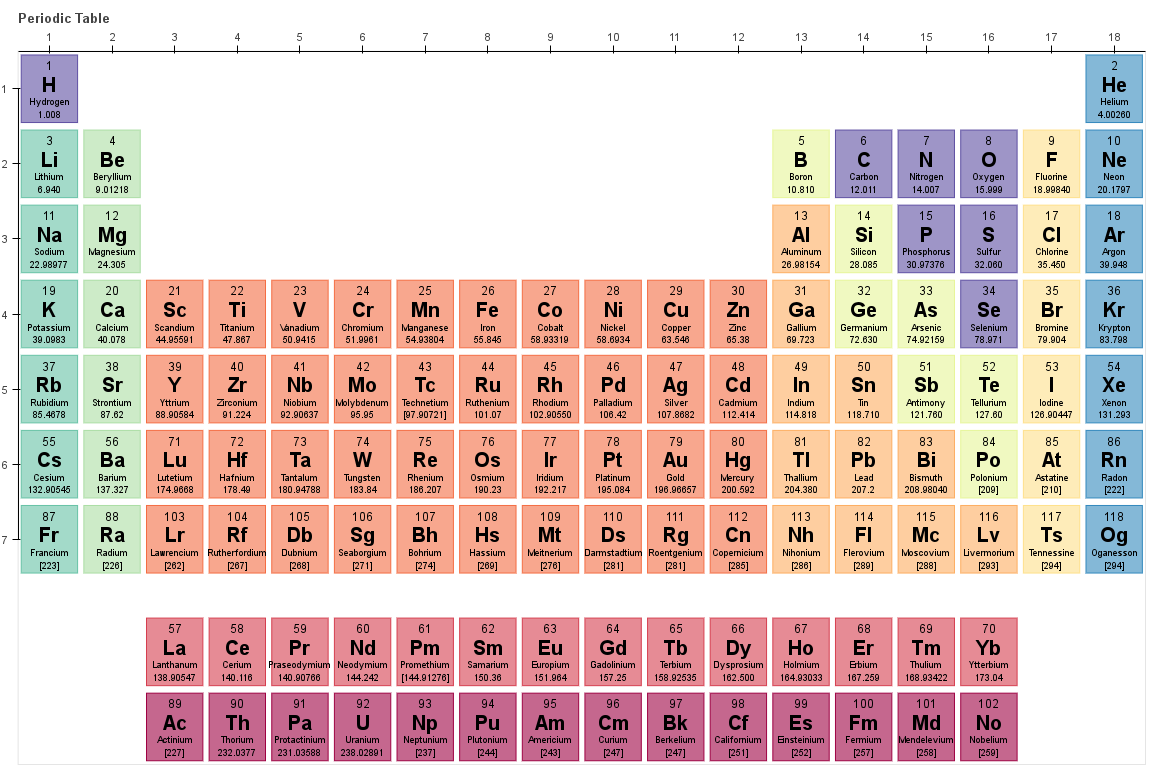
His periodic table was a significant breakthrough, providing a framework for understanding the relationships between elements. Mendeleev left gaps in his table for elements that were yet to be discovered, accurately predicting their properties based on the trends he observed. His table consisted of elements arranged in order of increasing atomic mass, with elements having similar properties placed in the same vertical columns. Mendeleev's Periodic Tableĭmitri Mendeleev, a Russian chemist, is widely credited with the creation of the first periodic table in 1869. This article aims to compare the attributes of Mendeleev's periodic table with the modern periodic table, highlighting their similarities and differences. The development of the periodic table has evolved over time, with Dmitri Mendeleev's original version in the 19th century serving as the foundation for the modern periodic table we use today. It provides a systematic way to understand and predict the behavior of elements. The periodic table is a fundamental tool in chemistry that organizes elements based on their atomic number, electron configuration, and recurring chemical properties.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
